Impact of Different Fumed Silica on the Thickening and Thixotropic Properties of Polyurethane Adhesives
Polyurethane adhesives are widely used in the automotive, construction, electronics, and packaging industries. However, polyurethane adhesives often suffer from problems such as sagging and poor workability, especially when bonding vertical surfaces or complex structures. To address these issues, thickeners and thixotropic agents have become key methods for modification.
Fumed silica, with its unique high specific surface area, high surface activity, and three-dimensional network structure, is widely used to regulate the rheological properties of adhesives, achieving the ideal thixotropic behavior of “high viscosity under low shear and low viscosity under high shear.”
HIFULL experts studied the effects of different grades of fumed silica on the thickening and thixotropic properties of polyurethane adhesive components A and B. They systematically compared the differences in thickening viscosity and thixotropic values of HB-139, HB-139B, other brands, and HL-200.
Figure 1
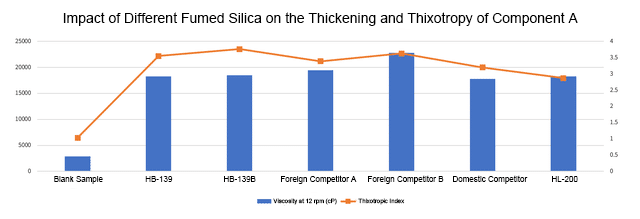
As shown in Figure 1, in component A, the blank sample’s viscosity at 12 rpm was approximately 2500 cP, indicating that the system has extremely weak thickening ability without the addition of fillers.
After adding HB-139, the viscosity increased to approximately 18000 cP; after adding HB-139B, the viscosity increased to approximately 18500 cP; after adding silica from brand A, the viscosity increased to approximately 19500 cP; after adding silica from brand B, the viscosity increased to approximately 22500 cP; after adding a product from competitor C, the viscosity increased to approximately 17800 cP; and after adding HL-200, the viscosity increased to approximately 18200 cP.
In terms of thixotropic value, the blank sample has a value of about 1.1, showing almost no thixotropy and poor shear-thinning effect. The thixotropic value of HB-139 jumped to about 3.5, HB-139B to about 3.7, competitor A to about 3.4, competitor B to about 3.6, the domestic competitor to about 3.0, and HL-200 to about 2.8.
In terms of viscosity performance, competitor B showed the highest viscosity among all samples in Component A, with outstanding thickening properties, while competitor C showed weaker thickening performance. HB-139 and HB-139B had thickening effects comparable to other grades. Regarding thixotropic performance, HB-139B had the highest value among all samples in Component A, indicating that the destruction and recovery efficiency of the three-dimensional network it constructs under shear action is optimal.
Figure 2
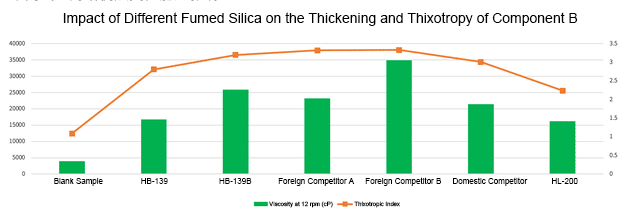
As shown in Figure 2, in Component B, the blank sample’s viscosity at 12 rpm is approximately 4000 cP, indicating insufficient thickening. The viscosity increased to about 16500 cP with HB-139, about 26000 cP with HB-139B, about 23000 cP with competitor A, about 35000 cP with foreign competitor B, about 21000 cP with competitor C, and about 16000 cP with HL-200.
In terms of thixotropic value, the blank sample has a value of about 1.2, lacking thixotropy. The value jumped to about 2.8 with HB-139, about 3.2 with HB-139B, about 3.3 with foreign competitor A, about 3.3 with competitor B, about 3.0 with competitor C, and about 2.2 with HL-200.
HB-139B achieved the best balance of “high viscosity + high thixotropy” in Component B. Its viscosity increase was more than 6 times that of the blank, and the thixotropic value was stable at 3.1, indicating that it can provide excellent sag resistance in a static state, while rapidly reducing viscosity during stirring or coating to facilitate construction.
The excellent performance of HB-139 and HB-139B stems from the compatibility of their surface modification technology with the polyurethane system.
The interaction between the surface hydroxyl groups of fumed silica and the polyurethane molecular chains (formation and destruction of hydrogen bonds) is the core mechanism for constructing the thixotropic network.
HB-139 and HB-139B utilize surface treatment processes better suited for polyurethane, resulting in higher entanglement efficiency between their surface active groups and polyurethane molecular chains. Thus, efficient thickening and thixotropic control can be achieved at lower addition levels. Furthermore, their superior dispersion of nanoparticles avoids performance degradation caused by agglomeration, further ensuring the stability and consistency of the system.
HB-139B demonstrates more excellent thixotropic control capability, more efficient thickening efficiency with low addition levels, superior dispersion stability, and better environmental friendliness and compatibility in polyurethane adhesives. It is evident that in the future, the development of fumed silica in the field of polyurethane adhesives will continue to move towards high performance, customization, functionality, and green development.
In terms of high performance and customization, by precisely controlling the specific surface area, surface chemical composition, and particle size distribution of fumed silica, customized designs for the thickening and thixotropic properties of polyurethane adhesives can be achieved, meeting the demand for “extreme processability” in high-end fields such as automotive lightweighting and precision electronic packaging. For example, in polyurethane adhesives for wind power blades, ultra-high-efficiency thixotropic fumed silica can be developed to ensure the adhesive does not sag during application on complex curved surfaces while achieving uniform coating.